Helium: ke revisions ke biich ke antar
Content deleted Content added
m r2.7.2) (robot Modifying: be-x-old:Гель |
No edit summary |
||
| Rekha 30: | Rekha 30: | ||
[[File:Goodyear-blimp.jpg|thumb|right|Because it is very light, helium is the gas of choice to fill [[airship]]s such as the [[Goodyear blimp]]]] |
[[File:Goodyear-blimp.jpg|thumb|right|Because it is very light, helium is the gas of choice to fill [[airship]]s such as the [[Goodyear blimp]]]] |
||
'''Helium''' ek [[chemical element]] hae, jiske [[chemical symbol]] '''He''' aur [[atomic number]] 2 hae. |
'''Helium''' ek [[chemical element]] hae, jiske [[chemical symbol]] '''He''' aur [[atomic number]] 2 hae. |
||
Ii bina rang ke ek gas hae, jon ki dusra element re react nai hoe hae. Iske halluk hoe ke kaaran, iske baloon me kaam e lawa jaawe hae. |
|||
== Chhapa ke gallery == |
== Chhapa ke gallery == |
||
11:18, 2 August 2011 ke badlao
| Atomic Structure | |
|---|---|
| Symbol | He |
| Atomic Number | 2 |
| Atomic Mass | 4.002602 g/mol |
| Periodic Table | |
| Group | 8 |
| Row / Period | 1 |
| Element Category | Noble gas |
| Chhapa | |
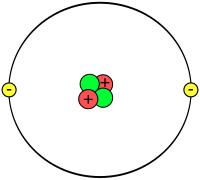 | |

Helium ek chemical element hae, jiske chemical symbol He aur atomic number 2 hae. Ii bina rang ke ek gas hae, jon ki dusra element re react nai hoe hae. Iske halluk hoe ke kaaran, iske baloon me kaam e lawa jaawe hae.
Chhapa ke gallery
-
Superfluidity
-
Diagram of He-4
-
Why He is unbound
-
Discharge tube
-
Glowing helium
| Ii vigyan article ek chhota panna hae. Aap iske lamba karke Wikipedia ke madat kare saktaa hae. |
Periodic table
| |||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| H | He | ||||||||||||||||||||||||||||||||||||||||
| Li | Be | B | C | N | O | F | Ne | ||||||||||||||||||||||||||||||||||
| Na | Mg | Al | Si | P | S | Cl | Ar | ||||||||||||||||||||||||||||||||||
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr | ||||||||||||||||||||||||
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe | ||||||||||||||||||||||||
| Cs | Ba | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn | ||||||||||
| Fr | Ra | Ac | Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og | ||||||||||
| |||||||||||||||||||||||||||||||||||||||||
Template:Link FA Template:Link FA Template:Link FA Template:Link FA







